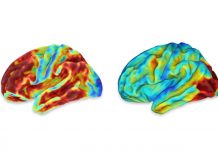
Newswise — The federal Food and Drug Administration’s recent approval of aducanumab, a treatment for Alzheimer’s disease, has drawn decidedly mixed reviews.
Advocates for patients, such as the Alzheimer’s Association, applauded the FDA’s green light for the drug (though the group later decried the high price tag set by U.S. manufacturer Biogen). On the other side, critics assessed the trial data as inconclusive. Indeed, three of the scientists on the FDA advisory committee studying the data resigned in protest over the decision to approve aducanumab, which is also known by the brand name Aduhelm.
Johns Hopkins Carey Business School Senior Lecturer Supriya Munshaw – an expert in the commercialization of early-stage technologies, especially in the life science and medical device industries – has considered the recent controversy and offers her insights into the…