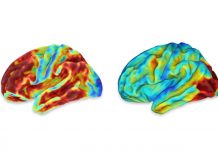
After years of disappointing studies of potential treatments for the more than 6 million people living with Alzheimer’s disease, the U.S. Food and Drug Administration recently approved a new drug called aducanumab that is sure to garner significant interest.
The drug, however, followed an unusual regulatory path and did not convince a key advisory panel that it works.
Dr. G. Caleb Alexander, an internist and a professor of epidemiology and medicine in the Johns Hopkins Bloomberg School of Public Health, served on that committee and spoke with The Baltimore Sun about the new drug.
So why all the buzz about this drug?
There is no cure for…